Myelin, a lipid-rich membrane sheath surrounding some axons, insulates against the flow of ions. 
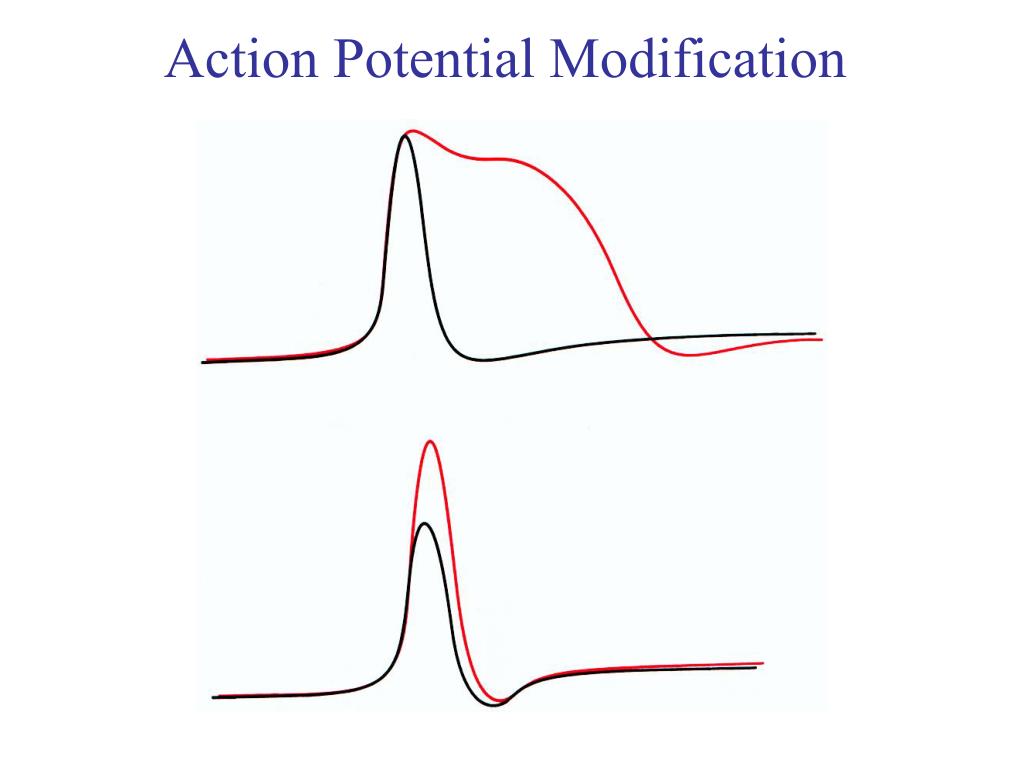
The brief dip in the membrane potential below the normal resting voltage is termed hyperpolarization.Īction potentials propagate a signal along the length of an axon differently in myelinated versus unmyelinated axons. However, the Kv have slow kinetics and remain open slightly longer than needed to return the cell to resting membrane voltage. When the membrane potential falls below the threshold, both the Nav and the Kv begin to close. The flow of potassium ions out of the cell results in a decrease in membrane potential towards the cell’s resting voltage. Therefore, after approximately 1 msec, there is an opening of the slower Kv channels that is coincident with the inactivation of the faster Nav channels. Although Kv have approximately the same threshold voltage as Na, the kinetics of the potassium channel are much slower. Repolarization begins as voltage-gated potassium channels (Kv) open. Depolarization in mature neurons lasts approximately 1 msec, at which time the Nav are inactivated and no longer able to flux ions. The flow of positive sodium ions into the cell leads to further depolarization of the membrane, thus opening more Nav in a positive-feedback loop. The initial depolarization is determined by the cell’s threshold voltage, the membrane potential at which voltage-gated sodium channels (Nav) open to allow an influx of sodium ions. Ī neuronal action potential has three main stages: depolarization, repolarization, and hyperpolarization. The thinking is that this clustering optimizes action potential conduction and fidelity by lowering the number of channels required for propagation and by increasing the speed of conduction, compared to diffuse channel localization. Similarly, there is clustering of voltage-gated sodium channels into lipid raft ‘micro-domains’ within unmyelinated axons. In myelinated axons, high-density clustering of voltage-gated channels to the nodes of Ranvier decreases the threshold for action potential initiation. In addition to increased receptor expression, the localization of voltage-gated ion channels is essential to efficient propagation of action potentials. By utilizing shorter action potentials, the cell can fire more rapidly and thus encode information more quickly. However, a developmental increase in sodium channel expression produces a more rapid depolarization, while a concurrent increase in potassium channels results in a shorter duration of action potentials. Įarly in development, action potentials are relatively slow-rising and elongated. Specifically, the decreased intracellular sodium concentration within mature neurons results in higher peak voltages of action potentials. Because the relative intracellular and extracellular concentrations of an ion determines the driving force of ions across the membrane, changes in ion concentration can significantly affect action potential dynamics. ĭuring embryonic development, the intracellular concentration of sodium significantly decreases. Additionally, the speed of action potential propagation along myelinated axons is increased throughout development as myelin thickens, and the distance between nodes of Ranvier lengthen. Below, we will specifically address changes in ion concentration, ion channel density, and ion channel location. There are several pre- and postnatal maturational processes that serve to modulate action potential formation and propagation. Similar to action potential initiation in neurons, and in contrast to pacemaker cells, myocardiocytes initiate rapid depolarization through voltage-gated sodium channels. The action potential is then dispersed throughout the heart by myocardiocytes, cardiac muscle cells that contract while they conduct the current to neighboring cells. A transient current of calcium ions, mediated by T-type calcium channels, slowly depolarizes the pacemaker cell until reaching the threshold potential for L-type voltage-gated calcium channels, inducing an action potential. Unlike in neurons, the majority of current in pacemaker cells gets mediated through calcium flux. Within the heart, pacemaker cells located in the SA node initiate action potentials intrinsically and rhythmically. These variables are directly dependent upon the number, location, and kinetics of ion channels within the membrane. Within a population of neurons, there can be significant variability in the intrinsic electrical properties of the cell, such as resting potential, maximum firing rate, resistance to current, and width of action potentials. Although usually discussed in the context of neuronal cells, action potentials also occur in many excitable cells such as cardiac muscle and some endocrine cells.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
